Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry . what is the sublimation process? Sublimation, including melting, freezing, and evaporation, is a form of.
from chemistnotes.com
what is the sublimation process? Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas. Sublimation, including melting, freezing, and evaporation, is a form of.
Sublimation Definition/Meaning Chemistry Notes
Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Explain the relation between phase transition temperatures and intermolecular attractive forces.finally, sublimation is the process in which a solid is change to a gas.the reverse of sublimation is called deposition, a process in which gaseous substances condense directly into the solid state, bypassing the liquid state. The formation of frost is an example of.
From www.researchgate.net
Schematic view of what happens in a fusionevaporation reaction. In Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Its complement, deposition , is defined as a conversion. The formation of frost is an example of.finally, sublimation is the process in which a solid is change to a gas. Solid co 2, also known as dry ice, goes directly from a solid phase to a gaseous phase in a process called sublimation. The enthalpy of sublimation, δhsub,. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From judith-kconner.blogspot.com
Explain the Difference Between Evaporation and Sublimation Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistrylike vaporization, the process of sublimation requires an input of energy to overcome intermolecular attractions.finally, sublimation is the process in which a solid is change to a gas. Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas. Define phase transitions and phase transition temperatures. The formation of frost is an example. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From studiousguy.com
7 Sublimation Examples in Daily Life StudiousGuy Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Define phase transitions and phase transition temperatures. Sublimation, including melting, freezing, and evaporation, is a form of. Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas. what is the sublimation process? The enthalpy of sublimation, δhsub, is the energy required to convert one mole of a substance from the. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From data.allenai.org
changes of state (lesson 0771) TQA explorer Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry The reverse process of sublimation is called deposition, a process.finally, sublimation is the process in which a solid is change to a gas. The enthalpy of sublimation, δhsub, is the energy required to convert one mole of a substance from the. The formation of frost is an example of.like vaporization, the process of sublimation requires an. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From app.emaze.com
Science Molecules on emaze Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistrythe reverse of sublimation is called deposition, a process in which gaseous substances condense directly into the solid state, bypassing the liquid state.finally, sublimation is the process in which a solid is change to a gas. Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas. Sublimation, including melting, freezing, and evaporation,. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From apollo.nvu.vsc.edu
Latent Heat of evaporation, fusion, and freezing Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Define phase transitions and phase transition temperatures. Explain the relation between phase transition temperatures and intermolecular attractive forces.like vaporization, the process of sublimation requires an input of energy to overcome intermolecular attractions. Solid co 2, also known as dry ice, goes directly from a solid phase to a gaseous phase in a process called sublimation. Sublimation, including melting,. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From stacklima.com
Qu’estce que la vaporisation ? StackLima Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry The formation of frost is an example of. Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas.like vaporization, the process of sublimation requires an input of energy to overcome intermolecular attractions. Define phase transitions and phase transition temperatures. Its complement, deposition , is defined as a conversion. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
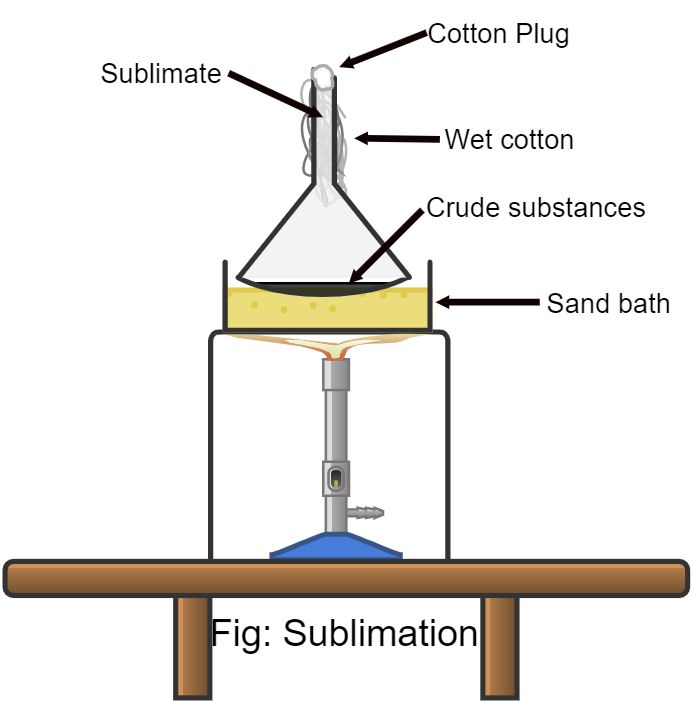
From www.teachoo.com
What is Sublimation? 5+ Examples with Diagram Teachoo Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Solid co 2, also known as dry ice, goes directly from a solid phase to a gaseous phase in a process called sublimation.finally, sublimation is the process in which a solid is change to a gas. Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas. Its complement, deposition , is defined as. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From www.youtube.com
What is sublimation fusion and why evaporation causes cooling class 9th Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Define phase transitions and phase transition temperatures. The reverse process of sublimation is called deposition, a process. The enthalpy of sublimation, δhsub, is the energy required to convert one mole of a substance from the. Sublimation, including melting, freezing, and evaporation, is a form of. Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From www.youtube.com
Change of State Evaporation Condensation Melting Freezing Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Define phase transitions and phase transition temperatures. The reverse process of sublimation is called deposition, a process. Explain the relation between phase transition temperatures and intermolecular attractive forces.finally, sublimation is the process in which a solid is change to a gas. The formation of frost is an example of. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From www.youtube.com
Changing states of matter 🔁 Melting, Freezing, Evaporation Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry The reverse process of sublimation is called deposition, a process. what is the sublimation process?finally, sublimation is the process in which a solid is change to a gas.the reverse of sublimation is called deposition, a process in which gaseous substances condense directly into the solid state, bypassing the liquid state. Explain the relation between phase. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From www.youtube.com
Science 9 Chapter 2 Evaporation sublimation, Chromatography Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistrylike vaporization, the process of sublimation requires an input of energy to overcome intermolecular attractions. The enthalpy of sublimation, δhsub, is the energy required to convert one mole of a substance from the. Sublimation, including melting, freezing, and evaporation, is a form of.the reverse of sublimation is called deposition, a process in which gaseous substances condense directly. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From www.teachoo.com
For any substance, why does the temperature remain constant during the Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Explain the relation between phase transition temperatures and intermolecular attractive forces. Solid co 2, also known as dry ice, goes directly from a solid phase to a gaseous phase in a process called sublimation. Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas. Its complement, deposition , is defined as a conversion. The reverse. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From blogdejuanmateacher.blogspot.com
El blog de juanmateacher 6º CHANGES OF STATE OF MATTER Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry The reverse process of sublimation is called deposition, a process. what is the sublimation process? Its complement, deposition , is defined as a conversion.the reverse of sublimation is called deposition, a process in which gaseous substances condense directly into the solid state, bypassing the liquid state. The formation of frost is an example of. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From www.chemistrylearner.com
Heat (Enthalpy) of Fusion Definition, Equation, and Problems Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Explain the relation between phase transition temperatures and intermolecular attractive forces. Sublimation, including melting, freezing, and evaporation, is a form of. what is the sublimation process? Define phase transitions and phase transition temperatures. Its complement, deposition , is defined as a conversion. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From www.expii.com
Sublimation — Definition & Overview Expii Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Define phase transitions and phase transition temperatures.the reverse of sublimation is called deposition, a process in which gaseous substances condense directly into the solid state, bypassing the liquid state. Dry ice undergoes sublimation to change from solid carbon dioxide directly into carbon dioxide gas. The enthalpy of sublimation, δhsub, is the energy required to convert one mole of. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From www.researchgate.net
a) Compiled experimental ER cross sections of fusionevaporation Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry The formation of frost is an example of.finally, sublimation is the process in which a solid is change to a gas.like vaporization, the process of sublimation requires an input of energy to overcome intermolecular attractions.the reverse of sublimation is called deposition, a process in which gaseous substances condense directly into the solid state, bypassing. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.
From smartclass4kids.com
Changing States of Matter Solid, Liquid,Gas, Phase Change Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry Sublimation, including melting, freezing, and evaporation, is a form of.like vaporization, the process of sublimation requires an input of energy to overcome intermolecular attractions. The formation of frost is an example of. Define phase transitions and phase transition temperatures.finally, sublimation is the process in which a solid is change to a gas. Explain The Process Of Fusion Evaporation And Sublimation In Aspects Of Chemistry.